Battery model justification
Lead-Acid batteries
We have abandoned classical models (such as Shepherd's model1), which involve numerous parameters typically requiring detailed measurements for each battery type.
We have developed a relatively simple model that reproduces fundamental data provided by all manufacturers, based on information typically found in standard datasheets.
We have incorporated specific behaviors not detailed in datasheets. When developing the model, we worked closely with a manufacturer's technical team. They provided insights and specific data presumed to have general validity for all lead-acid batteries based on their common chemistry, such as self-discharge values and temperature behavior, capacity temperature dependence, etc. Other publications also provided valuable information 2, 3, 4, 5.
For these secondary behaviors, when unknown, the user can do with the default values, specific to each type of technology, and proposed by the software.
Li-Ion batteries
Li-ion battery operating parameters and performance are not publicly available. When developing the model, very few manufacturers were willing to share detailed operating data for their products, and such data was typically confidential.
We developed a model that best reproduces the behavior shown by the curves we obtained. In fact, the model developed for lead-acid batteries could be adapted without significant difficulty. Parameters were adjusted to match sets of curves such as voltage discharge curves (and potentially other curves not shown here).
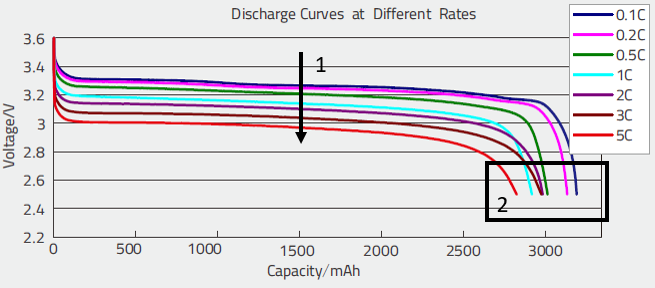
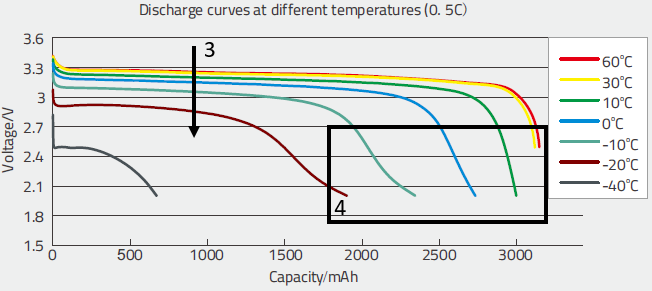
These curves allowed to evaluate:
- The basic voltage at SOC = 0.5, and slope as a function of the SOC, for the evaluation of VocBattery.
- The behaviour at high SOC (overcharging) and deep discharge,
- The drop of voltage as a function of the discharge rate (1), which allows namely to evaluate the internal resistance,
- The diminution of the capacity as a function of the discharge rate (2); this allows to evaluate the parameter of the Peukert model.
- The diminution of the voltage when the temperature decreases (3), this shows that the variation becomes more significant below 20°C,
- The huge diminution of the capacity with low temperatures (4). Again, the effect is negligible above 30°C and becomes significant below 20°C.
All of these parameters may be different for different chemistries, but we admitted that they remain rather homogeneous within a same technology. We did not obtain recent detailed data (likely increasingly confidential), so we typically apply the same model for secondary parameters in modern batteries, for which we have limited information.
Therefore when defining a new battery, we use the parameters of an existing cell of the same technology, and modify only the parameters available in the datasheets.
-
C .M. Shepherd
Design of Primary and Secondary Cells.
and:
An equation describing Battery Discharge.
Journal of the Electrochemical Society, vol 112(7), July 1965, pp 657-664. ↩ -
Watsun-PV - User's Manual and Program Documentation
Watsun Simulation Laboratory, University of Waterloo, Waterloo, Ontario N2L 3G1, 1992. ↩ -
Bopp, Gabler, Sauer, Jossen, Höhe, Mittermeier, Bächler,Sprau, Willer, Wollny.
A Systematic Effort de Define Evaluation and Performance Parameters and Criteria for Lead-acid Batteries in PV Systems.
13th European Photovoltaic Solar Energy Conference, Nice, Oct 1995. ↩ -
J.B. Copetti, F. Chenlo and E. Lorenzo
Comparison between Charge and Discharge Battery Models and Real Data for PV Applications.
11th European Photovoltaic Solar Energy Conference, Montreux, Oct 1992, pp1131-1134. ↩ -
J.B. Copetti, F. Chenlo
Internal Resistance Characterization of Lead-Acid Batteries for PV Rates
11th European Photovoltaic Solar Energy Conference, Montreux, Oct 1992, pp1116-1119. ↩