Capacity as function of the discharge rate
The capacity is highly varying according to the charge/discharge rate, i.e. the current.
NB: The discharge rate [hours] is the time for fully discharging the battery at a given current. In PVsyst, the nominal capacity is specified at C10, i.e a discharge in 10 hours. This corresponds to a current of 0.1 times the capacity, sometimes noted 0.1C.
Peukert model
To compute the battery capacity at any current, PVsyst uses Peukert's model. The capacity is corrected according to the discharge rate and the peukert coefficient K, specified in the battery's parameters (usual value K = 1.02 for Li-ion, 1.12 to 1.13 for Lead Acid):
or
Where:
- CapNom [Ah] = nominal capacity, defined for the reference discharge time
- DischRate(CapNom) [hours] = reference discharge time (usually 10 hours)
- Current(CapNom) [A] = CapNom [Ah] / DischRate(CapNom) [hours]
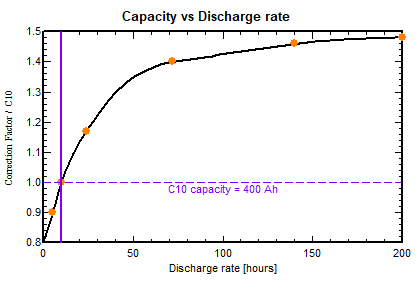
Lead-acid batteries
The capacity variation is very important: we observe a ratio of about 40 % between C10 and C100 ! In PVsyst, this behaviour is defined as a profile.
This will have deep consequences on the battery balance and efficiency when using the battery in a simulation.
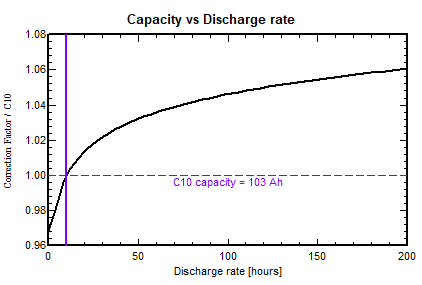
Lithium-Ion batteries
The capacity variation is less pronounced. The capacity increase at C100 is about 4%. The capacity variation is less pronounced. The capacity increase at C100 is about 4%.
Peukert model limitations
Peukert's model is based on tests done on batteries that were discharged at constant current, until they reach a low voltage threshold, or until the BMS stops the discharge.
But this condition does not mean the battery is completely empty, only that is it not possible to continue discharging the battery at the current rate.
The reason why the battery stopped discharging is a mix of electrical and chemical phenomenons. Some are non reversible (ohmic losses), some are near instantaneous (voltage drop to the internal resistance) amd some are reversible after some time (chemical effects).
This has two consequences :
- The capacity reduction at higher discharge rate must not be interpreted as a loss
- A higher discharge rate will trigger the end of the discharge earlier, even when the battery still contains a significant energy.
This effect will be used in the SOC evaluation.